Ion-selective electrodes
prepared by Wojciech Wroblewski
Chemical sensors are miniaturized analytical devices, which can deliver real-time and on-line information on the presence of specific compounds or ions in complex samples. Usually an analyte recognition process takes place followed by the conversion of chemical information into an electrical or optical signal. Among various classes of chemical sensorsion-selective electrodes (ISE) are one of the most frequently used potentiometric sensors during laboratory analysis as well as in industry, process control, physiological measurements, and environmental monitoring. The principle of ion-selective electrodes operation is quite well investigated and understood.
An ion-selective membrane is the key component of all potentiometric ion sensors. It establishes the preference with which the sensor responds to the analyte in the presence of various interfering ions from the sample. If ions can penetrate the boundary between two phases, then an electrochemical equilibrium will be reached, in which different potentials in the two phases are formed. If only one type of an ion can be exchanged between the two phases, then the potential difference formed between the phases is governed only by the activities of this target ion in these phases. When the membrane separates two solutions of different ionic activities (a1 and a2) and provided the membrane is only permeable to this single type of ion, the potential difference (E) across the membrane is described by the Nernst equation:
E = RT/zF · ln (a2/a1)
If the activity of the target ion in phase 1 is kept constant, the unknown activity in phase 2 (a1 = ax) is related to (E) by:
E = RT/zxF · ln (ax/a1) = const + S · log (ax)
where S=59.16/z [mV] at 298 K and zx - the charge of the analyte. The potential difference can be measured between two identical reference electrodes placed in the two phases. In practice the potential difference i.e. the electromotive force is measured between an ion selective electrode and a reference electrode, placed in the sample solution. An examplary set-up for the measurement of electromotive force is presented in figure 1. It is important to note that this is a measurement at zero current i.e. under equilibrium conditions. Equilibrium means that the transfer of ions from the membrane into solution is equal to the transfer from the solution to the membrane. The measured signal is the sum of different potentials generated at all solid-solid, solid-liquid and liquid-liquid interfaces.
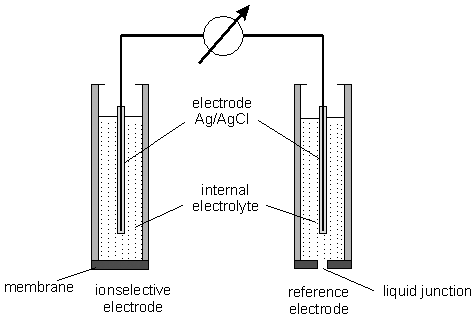
Fig.1 Measurement set-up of ion selectiveelectrode (ISE)
Using a series of calibrating solutions the response curve or calibration curve of an ion-selective electrode can be measured and plotted as the signal (electromotive force) versus the activity of the analyte. Typical calibration curve of a potentiometric sensor determined in this way is shown in figure 2. The linear range of the calibration curve is usually applied to determine the activity of the target ion in any unknown solution. However, it should be pointed out that only at constant ionic strength a linear relationship between the signal measured and the concentration of the analyte is maintained (because of the clear cut relationship between ion activity and concentration, occurring in such condition).
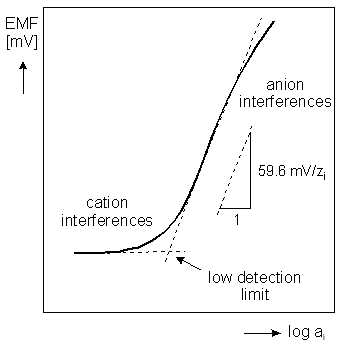
Fig.2 Typical calibration curve of an ion-selective electrode
Ions, present in the sample, for which the membrane is non-permeable (i.e. non-selective), will have no effect on the mesured potential difference. However, a membrane truly selective for a single type of an ion and completely non-selective for other ions does not exist. For this reason the potential of such a membrane is governed mainly by the activity of the primary (target) ion and also by the activity of other secondary (interfering) ions. The influence of the presence of interfering species in a sample solution on the measured potential difference is taken into consideration in the Nikolski-Eisenman formalism:
E = const + S · (log (ax) + (zx/zy) · log (Kxy · ay))
where (ay) is the activity of an interfering ion, (zy) its charge and (Kxy) the selectivity coefficient (determined empirically).
Characterization of an ion-selective electrode
The properties of an ion-selective electrode are characterised by parameters like:
- Selectivity. The selectivity is one of the most important characteristics of an electrode, as it oftendetermines whether a reliable measurement in the sample is possible or not. The selectivity coefficient (Kxy) has been introducedin the Nikolski-Eisenman equation. Most often it is expressed as the logarithm of (Kxy). Negative values indicate a preference for the target ion relative to the interfering ion. Positive values of log Kxy indicate the preference of an electrode for theinterfering ion. The experimental selectivity coefficients depend on the activity and a method of their determination.Different methods of the selectivity determination can be found in the literature. The IUPAC suggests two methods: separate solution method (SSM) and fixed interference method (FIM). There is also an alternative method of the selectivity determination called matchedpotential method (MPM). Each of them has got advantages and drawbacks, and there are not general rules pointing which method gives the true result. The methods proposed by IUPAC with several precautions will give meaningful data.
- Slope of the linear part of the measured calibration curve of the electrode. The theoretical valueaccording to the Nernst equation is: 59.16 [mV/log(ax)]at 298 K for a single charged ion or 59.16/2 = 29.58 [mV perdecade] for a double charged ion. A useful slope can be regarded as 50-60 [mV per decade] (25-30 [mV per decade] for double charged ion respectively). However, in certain applications the value of the electrode slope is not criticaland worse value does not exclude its usefulness.
- Range of linear response. At high and very low target ion activities there are deviations from linearity.Typically, the electrode calibration curve exhibits linear response range between 10-1M and 10-5M.
- Detection limit. According the IUPAC recommendation the detection limit is defined by the cross-sectionof the two extrapolated linear parts od the ion-selective calibration curve. In practice, detection limit on the order of 10-5-10-6M is measured for most of ion-selective electrodes. The observed detection limit is often governedby the presence of other interfering ions or impurities. If for example metal buffers are used to eliminate the effectswhich lead to the contamination of very dilute solutions it is possible to enhance the detection limit down to 10-10M.
- Response time. In earlier IUPAC recommendations, it was defined as the time between the instant atwhich the ion-selective electrode and a reference electrode are dipped in the sample solution (or the time at which the ion concentration in a solution is changed on contact with ISE and a reference electrode) and the first instant at which the potentialof the cell becomes equal to its steady-state value within 1 [mV] or has reached 90% of the final value (in certain cases also 63% or 95%).This definition can be extended to consider the drift of the system. In this case, the second time instant is defined as the one at whichthe EMF/time slope becomes equal to a limiting value. However, it should be pointed out that a single time constant does not describe the form of the electrode response function. Moreover, in many investigationsthe response time of the overall measuring system is determined, which influences on the response time of the ISE.
Construction of the ion-selective electrodes
Typical for all kinds of electrodes used in potentiometric ion measurements is a ion-sensitive membrane. This membrane can be prepared as:
- Solid membrane (e.g. glass membrane or crystal membrane)
- Liquid membrane (based on e.g. classical ion-exchanger, neutral or charged carrier)
- Membrane in a special electrode (gas-sensing or enzyme electrode). Typically such a membrane contains an analyte-selective component whichis responsible for the recognition process.
According to the nature of the binding sites, the membranes can be classified as: membranes containing fixed sites and membranes containing mobile ion-exchangers or ionophores (carriers). The binding sites are incorporated in the membrane matrix, which determines the internal polarity, lipophilicity, transport and other mechanical properties of the membrane.
Glass-membrane electrodes
The best known example of this type of electrodes is the glass electrode (pH electrode), in which the anionic fixed sites are created by defects in the SiO2 membrane and the cationic vacancies due to the nonsilicon constituents of glass. When the glass membrane is exposed to water a thick hydratedlayer is formed (5-100 nm), which exhibits improved mobility of the ions. The concentration of anionic binding is estimated between 3 and 10M, which determinethe wide linear range of the ISE calibration curve (typically 2-12 pH). The membrane is manufactured as a bulb of typical wall thickness of 0.05-0.2 mm (the optimum thickness is the result of compromise between the mechanical properties and electrical resistance). Two processes occur during the interaction of glass hydrated membrane and the sample solution (which both influence on the value of selectivity coefficient): ion-exchange and diffusion of all participating ions.
The example of an another glass-membrane electrode is the sodium-selective electrode (pNa electrode). The construction of this electrode is essentially identical to that of pH electrode, with the exception of the glass used (silica with 10% Na2O and Al2O3) and the fact that the inner reference solution has a fixed sodium ion activity. Interferences from hydrogen and silver cations are high, therefore the activity of such ions should be maintained some four order of magnitude less than the sodium ion activity to be measured. With proper precautions the range of measurable activities extends from 1 down to 10-8M. Although the pNa electrode is insensitive to ions other than H+ and Ag+, glass membranes selective for other ions (e.g.K+) have not been constructed.
Solid-state-membrane electrodes
Other types of membranes with fixed sites include single crystals of sparsely soluble salt and heterogeneous membranes in which the insoluble salt is incorporated in some suitable inert binder. In order to consider these layers at equilibrium it is necessary to use saturated solutions. In practice, these electrodes are applied in non-saturated solutions, so in this case the insoluble membrane slowly dissolves. Insoluble inorganic matererials as: Ag2S, CuS, CdS, PbS, LaF3, AgCl, AgBr, AgI and AgSCN have all been tested as cation exchange membranes, incorporated into an electrode body in the form of single crystal or compressed powder discs. These materials are ionic conductors, though the conductivity is extremely small and mainly takes place through the migration of point defects in the lattice. The response time of this membranes can be increased by incorporating aliovalent ions into the lattice (e.g. the fluoride-selective membrane LaF3 can be doped with Eu2+ ions). Sensors for the detection of: Ag+, Cu2+, Cd2+, Pb2+, S2-, F-, Br-, I-, SCN- and CN- ions can be constructed from such membranes. The sensitivity to ions of these electrodes arise from the dissolvation equilibria at the membrane surface.The measurement ranges of such electrodes lies in the range of 1-10-6M, but interference effects are frequently encountered.
It was also proved that it is possible to fabricate sensors by directly contacting the membrane with a wire (so called coated-wire electrodes) to form an ohmic contact. Such systems are found to exhibit complex behaviour (time and temperature dependences), requiring frequent re-calibration, but they are extremely simple to construct. An example is a silver-selective electrode, that has been designed by attaching a wire to the back of graphite/PTFE disc, on the front side of which the silver halide is rubbed in to the surface. Another exemplary coated-wire electrode was preparated by simply coating a wire with a membrane film of PVC containing an ion-exchanger.
Liquid-membrane electrodes
In addition to solid membranes, immiscible liquid (organic) phases with ion-exchange properties can be used, with such phases stabilised against the external solution phase within a polymer or ceramic membrane. The main component of electroactive membrane is neutral or charged compound, which is able to complex ions reversibly and to transfer them through an organic membrane by carrier translocation. This compound is called as an ionophore or an ion carrier. There are two kinds of ionophores: charged one (usually termed liquid exchanger) and neutralcarriers. They are mobile in both free and complexed forms, so the mobilities of all speciesare part of the selectivity coefficient together with ion-exchange equilibrium. The mobile binding sites are dissolved in a suitable solvent and usually trapped in a matrix of organic polymer (gel). Ion activity measurements are performed predominantly in aqueous media, so all membrane constituents are lipophilic. Therefore, the primary interaction between the ion in water and the lipophilic membrane containing the ionophore is the extraction process.
Typical polymeric membranes are based on plasticized poly(vinylchloride) (PVC) and containapproximately 66% of an plasticizer and 33% of PVC. Such a membrane is quite similar to liquid phase, because diffusion coefficients for dissolved low molecular weight ionophores are in the order of 10-7-10-8cm2/S. An appropriate plasticizer is added to a membrane in order to ensure the mobility of the free and complxed ionophore. It determines the membrane polarity and provides suitable mechanical properties of the membrane. The ionophore is usually present in 1% amount (approximately 10-2M), which is relatively low as compared to the glass electrode. A cation selective membrane can contain a salt of lipophylic anion and hydrophylic cation (additive), which improves performances of a membrane. Although other polymers like: polisiloxane, polystyrene, PMMA, polyamide or polyimide can be used as a membrane matrix, PVC is the most widely used matrix due to simplicity of membrane preparation.
Among the ion carriers, electrically neutral ionophores have found a wide field of applications as components in ion-selective liquid-membrane electrodes, e.g. in clinical chemistry, electrophysiology, as detectors in ion chromatography, in highly selective transport processes through artificial membranes (also biological membranes). As a results of the introduction of natural as well synthetic ionophores in ion-selective membranes, ISEs for direct measurement of various cations and anions were designed.
Modified-membrane electrodes
Additional selectivity can be attained by using composite membranes, in which an enzyme present in the outer part of the membrane catalyses a specific chemical reaction togenerate product ions. These ions can be detected by an internal ion-selective membrane. The well-known example is the selective detection of urea using urease as the enzyme catalyst. The ammonia generated, can then be detected by an ammonia or ammonium-selective electrodedescribed above. Similarly, enzyme reactions generating protons can be followed with glass or other proton-selective membranes. There is a multiplicity of enzyme-electrodes thatcan be made in this way, with substrates including aliphatic alcohols, acetylcholine, amygdalin, asparganine, glucose, glutamin, penicillin and other.
Modified electrode can be designed also as potentiometric gas sensors. The original concept was made for sensing carbon dioxide (Severinghaus electrode), but the principle on which this electrode operates is general for other gas sensors for the detection of: NH3, SO2, NO2, HCN etc. These electrodes are based on the measurement oflocal ion-cativity variation, caused by permeation of gas molecules (through a hydrophobicgas-permeable membrane) to the inner electrode compartment and their subsequent interaction with an internal solution. In the case of CO2 electrode the mechanism can be described by series of equilibrium: the partitioning of the gas molecules between the sample and the electrode (solubility equilibrium) and their hydrolysis inside the internal solution (e.g. 0.1M NaHCO3), which influences on the pH of this solution. The pH change is detected by an internal pH electrode (a bicarbonate-selective electrode may also be applied). It is important to note, that if the detected species is hydrogen ion, then all acid/base species will interfere. Improved selectivity is obtained by an appropriate choice of the internal electrode and by the differential gas permeability of the hydrophobic membrane.
|
Warsaw University of Technology Department of Analytical Chemistry Noakowskiego 3 00-664 Warsaw, Poland mail: brzozka@ch.pw.edu.pl phone: +48 22 234 5427 fax: +48 22 234 5631 www: csrg.ch.pw.edu.pl |


